Atom theory4/2/2024  
The law of definite composition says that the masses of elements that join to form a compound are always in the same ratio, or proportions.These were the basis for Dalton's theory. They discovered these laws when they measured how elements combined into compounds. Dalton and another chemist, Joseph Proust had discovered two laws: the law of definite composition and the law of multiple proportions. Chemical changes correspond to a reorganisation of the atoms taking part in the chemical reaction.ĭalton defined the atom as the basic unit of an element that can take part in a chemical combination.The atoms that make chemical compounds are present in set proportions.That atoms of the same element are of the same type and mass.That atoms are indivisible and invisible particles.In 1803, the English scientist John Dalton reworked Democritus' theory, as follows: In 1758, Roger Joseph Boscovich described a precursor of the atomic theory. They had no way to experiment to show whether his theory was true or not. Some other philosophers agreed, and others disagreed. Democritus thought that there was nothing between the atoms and that everything around us could be explained if we could understand how atoms worked. He thought that atoms would last forever, never change and could not be destroyed. Democritus called these small pieces of matter atoms, which means "indivisible". He said that this last piece of matter could not be cut any smaller. It does not apply in the same way to plasmas or neutron stars.ĭemocritus' atomic theory ĭemocritus thought that if you cut something in half again and again, you would at last have to stop. 
The theory applies to solids, liquids and gases. A lot of the ideas in the modern theory came from John Dalton, a British chemist and physicist. The first idea of the atom came from the Greek philosopher Democritus. These subatomic particles are made of quarks. However, it is now known that atoms are made of protons, neutrons, and electrons. Atoms were once thought to be the smallest pieces of matter. In chemistry and physics, the atomic theory explains how our understanding of the atom has changed over time. A Croatian Jesuit who provided a prototype of the atomic theory John Dalton (1766–1844), English chemist and physicist Sir Joseph John Thomson (1856–1940), English physicist, discovered the electron and its negative charge. This beam was deflected toward the positive charge and away from the negative charge, and was produced in the same way with identical properties when different metals were used for the electrodes.Democritus was a Greek philosopher, 460 BC Roger Joseph Boscovich. When high voltage was applied across the electrodes, a visible beam called a cathode ray appeared between them. This apparatus consisted of a sealed glass tube from which almost all the air had been removed the tube contained two metal electrodes. If matter were composed of atoms, what were atoms composed of? Were they the smallest particles, or was there something smaller? In the late 1800s, a number of scientists interested in questions like these investigated the electrical discharges that could be produced in low-pressure gases, with the most significant discovery made by English physicist J. While the historical persons and dates behind these experiments can be quite interesting, it is most important to understand the concepts resulting from their work. Here, we will discuss some of those key developments, with an emphasis on application of the scientific method, as well as understanding how the experimental evidence was analyzed. Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. And if atoms are neither created nor destroyed during a chemical change, then the total mass of matter present when matter changes from one type to another will remain constant (the law of conservation of matter). For example, if an element such as copper consists of only one kind of atom, then it cannot be broken down into simpler substances, that is, into substances composed of fewer types of atoms. 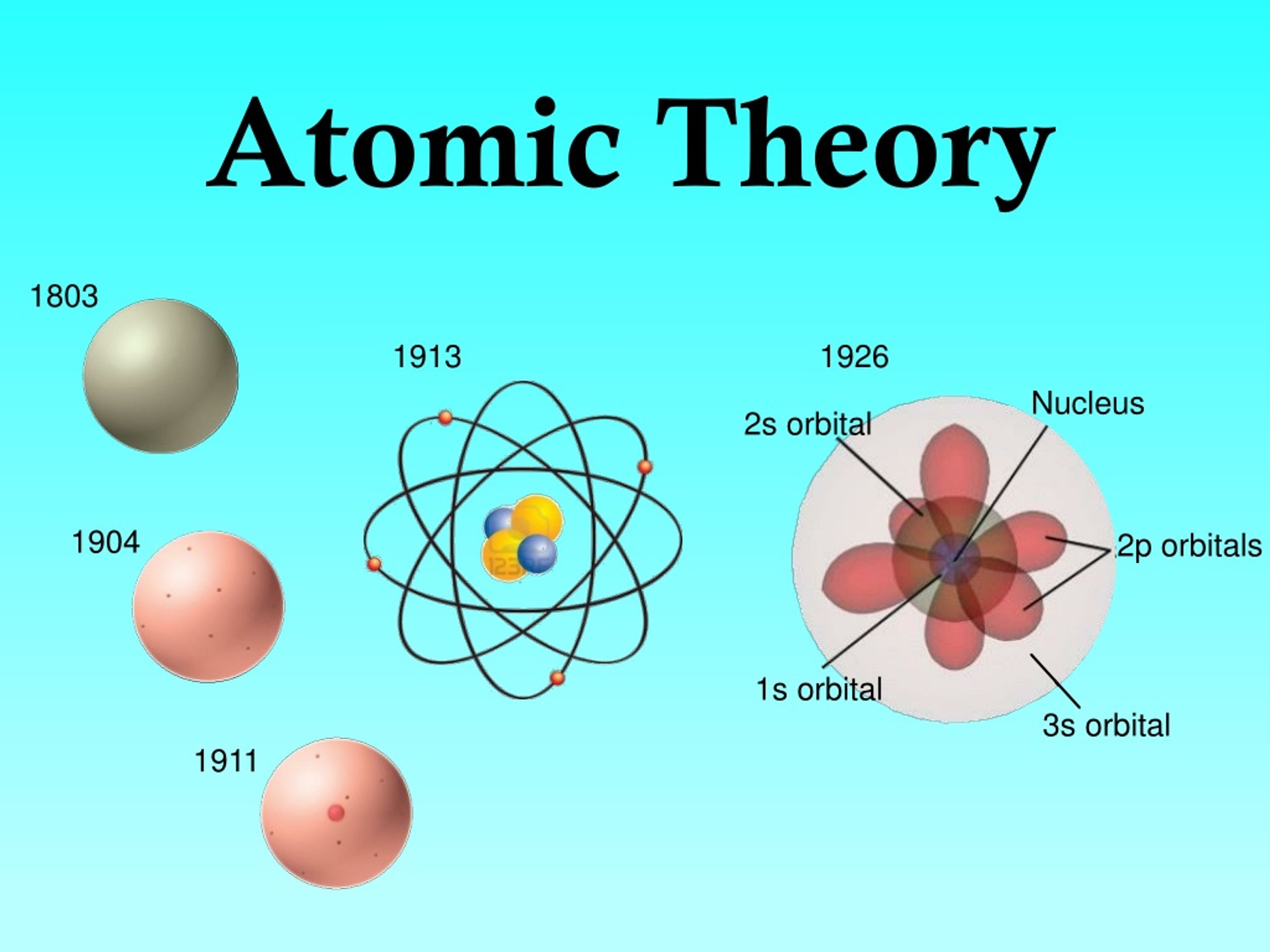
(credit copper: modification of work by ).ĭalton’s atomic theory provides a microscopic explanation of the many macroscopic properties of matter that you’ve learned about. \): When the elements copper (a shiny, red-brown solid, shown here as brown spheres) and oxygen (a clear and colorless gas, shown here as red spheres) react, their atoms rearrange to form a compound containing copper and oxygen (a powdery, black solid).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
